Chatper 7. Corrosion in metal and concrete (42 pages)
- 1.Corrosion in metal: local cell model
- 1.1.Anodic reactions and the tendency to corrosion
- 1.2.Cathodic reactions and corrosivity in water
- 1.2.1.Corrosion in iron in the absence of oxygen.
- 1.2.2.Corrosion in iron in the presence of dissolved oxygen
- 2.Formation of protective layers and passivation
- 2.1.Kinetics of the corrosion reactions
- 2.2.Natural water used in carbon steel passivation
- 2.3.High temperature passivating of iron using magnetite
- 2.4.Self-passivating alloys
- 3.Forms of corrosion
- 3.1.Generalised corrosion
- 3.1.1.Acid corrosion
- 3.1.2.Alkaline corrosion
- 3.1.3.Corrosion involving complexing agents
- 3.2.Localised corrosion
- 3.2.1.Differential aeration corrosion
- 3.2.2.Tuberculation
- 3.2.3.Galvanic corrosion
- 3.2.4.Pitting corrosion
- 3.2.5.Crevice corrosion
- 3.2.6.Stress corrosion
- 3.2.7.Intergranular corrosion
- 3.2.8.Erosion-corrosion
- 3.2.9.Cavitation corrosion
- 3.3.Microbiological corrosion
- 3.3.1.Sulphur reducing bacteria
- 3.3.2.Acidifying bacteria
- 3.3.3.Iron and manganese bacteria
- 3.3.4.Bacteria forming biofilms
- 3.3.5.Algae
- 3.3.6.Nitrifying bacteria
- 3.3.7.Identification
- 4.Corrosion in metallic materials
- 4.1.Corrosion in cast iron = graphitisation
- 4.2.Stainless steel
- 4.2.1.Definition and types
- 4.2.2.Different types of corrosion that affect stainless steels
- 4.2.3.Using stainless steels
- 4.3.Aluminium
- 4.4.Copper
- 4.5.Galvanised steel
- 4.6.Lead
- 4.7.Copper alloys
- 4.7.1.Brass
- 4.7.2.Cupro-nickel alloys
- 5.Secondary corrosion parameters
- 5.1.Impact made by mineralisation
- 5.1.1.Impact made by the overall mineralisation of water
- 5.1.2.The impact made by chlorides
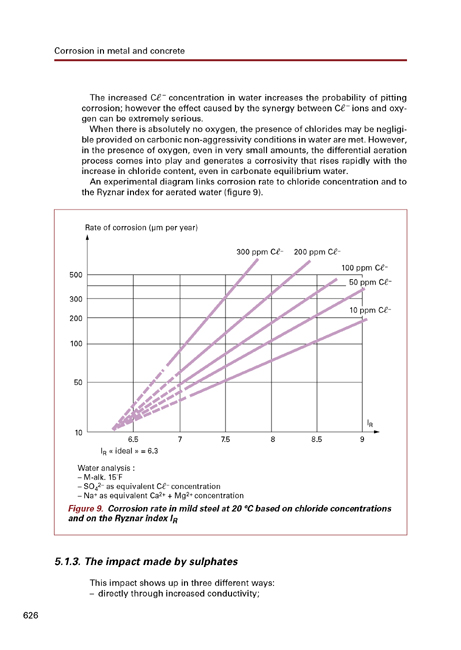
- 5.1.3.The impact made by sulphates
- 5.2.Effect of temperature on aerated water
- 5.2.1.Below 60 ºC
- 5.2.2.Above 60 ºC
- 5.3.Effect of surface condition and water cleanliness
- 5.4.Effect of circulation velocity
- 5.5.Effect of pH on aerated water
- 5.6.Effect of oxidising agents
- 5.6.1.Dissolved oxygen
- 5.6.2.Oxidising agents
- 5.7.Seawater
- 5.7.1.Protection against corrosion
- 5.7.2.Protection against soiling
- 6.Protection against corrosion
- 6.1.Corrosion inhibitors
- 6.1.1.Anodic inhibitors
- 6.1.2.Cathodic inhibitors
- 6.1.3.Combined anodic/cathodic inhibitors
- 6.1.4.Oxygen absorbers
- 6.1.5.Organic inhibitors
- 6.1.6.Protection for non-ferrous metals: copper and aluminium
- 6.1.7.The use of inhibitors in industrial circuits
- 6.2.Cathodic protection
- 7.Measuring the rate of corrosion
- 7.1.Introduction
- 7.2.Coupons
- 7.3.Test exchangers
- 7.4.Linear polarisation resistance (LPR) measurement
- 7.5.Chemical analysis
- 7.6.Circuit inspection
- 7.7.Results of an anticorrosion programme
- 8.Damage to concrete
- 8.1.Mechanical causes
- 8.2.Chemical causes
- 8.2.1.Carbonic attack: fresh water
- 8.2.2.Strong acid attack
- 8.2.3.Action of ammonium
- 8.2.4.Action of sulphates
- 8.2.5.Attack by strong bases (NaOH, KOH, Na2CO3)
- 8.2.6.Bacterial corrosion with the formation of H2S
Introduction
As water is transported or used, it can affect the various materials it comes into contact with in a number of different ways; the most frequent of these changes will take the form of corrosion affecting metal and, more particularly, steel. Other changes, affecting concrete, for instance, can also occur. It should be noted that in paragraphs 1, 2 and 3 describing the corrosion process and forms, almost all examples selected involve low alloy steels and that, although the same mechanisms apply, the reader is asked to refer to §4. for a description of behaviour with regard to corrosion in steel alloy and other materials: cast iron, aluminium, copper and their alloys.
© 2007